Where are hydrophobic amino acids located in cell membrane
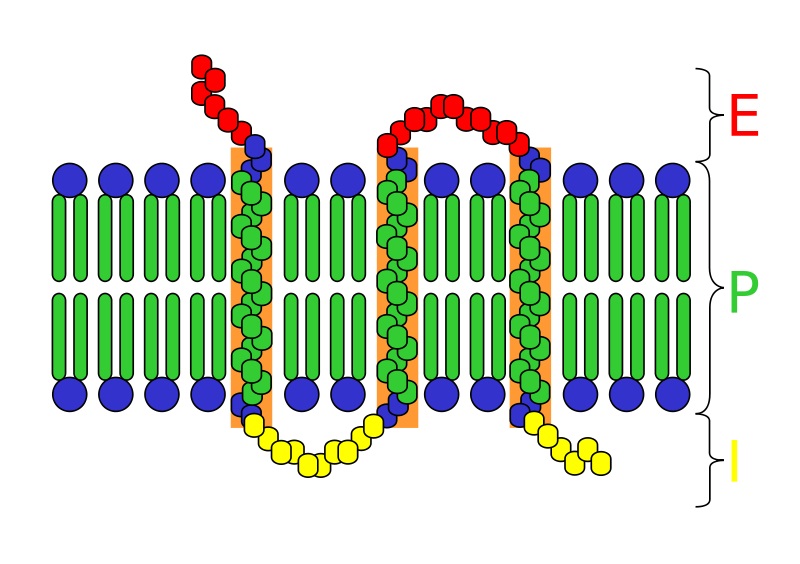
The hydrophobic or hydrophilic character of a compound or amino acid is sometimes called its hydropathic character, hydropathicity, or even “hydropathy” (which originally meant the therapeutic use of water). As expected, the trans-membrane -helices are generally devoid of polar amino acids while nonmembrane spanning portions of the protein are usually enriched in these amino acids. Hydrophobicity scales are routinely applied to the recognition of TM α-helices by averaging over a window and by searching for hydrophobic stretches at least 15 residues long. Since each amino acid in an -helix spans approximately 1.5, it takes approximately 20 amino acids to cross the hydrophobic interior (approximately 30 ). A hydrophobicity profile is a plot of the residue number versus residue hydrophobicity, averaged over a running window. When consecutively measuring amino acids of a protein, changes in value indicate attraction of specific protein regions towards the hydrophobic region inside the lipid bilayer. The hydrophilic head is composed of a choline structure (blue) and a. Hydrophobicity profiles have been used to predict the location of turns in proteins. Learn how they subdivide sections of a cell and how proteins in these membranes are. These scales are commonly used to predict the transmembrane alpha-helices of membrane proteins.
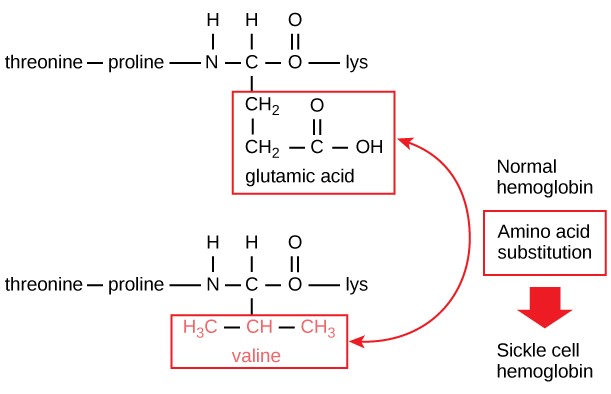
An inverted hydrophobicity scale corresponds to a hydrophobicity scale in which values are multiplied by 1.Ībout a dozen different scales of hydrophilicity or hydrophobicity have been derived from measurements of partition coefficients of amino acids between two noninteracting isotropic phases as well as by several other methods. Cell membranes are held together in part by hydrophobic effects. These residues are normally located inside the protein core, isolated from solvent. Usually, amino acids with non-polar residues will be found in the core of proteins. The more positive the value, the more hydrophobic are the amino acids located in that region of the protein. Hydrophobic amino acids The hydrophobic amino acids include alanine (Ala, A), valine (Val, V), leucine (Leu, L), isoleucine (Ile, I), proline (Pro, P), phenylalanine (Phe, F) and cysteine (Cys).
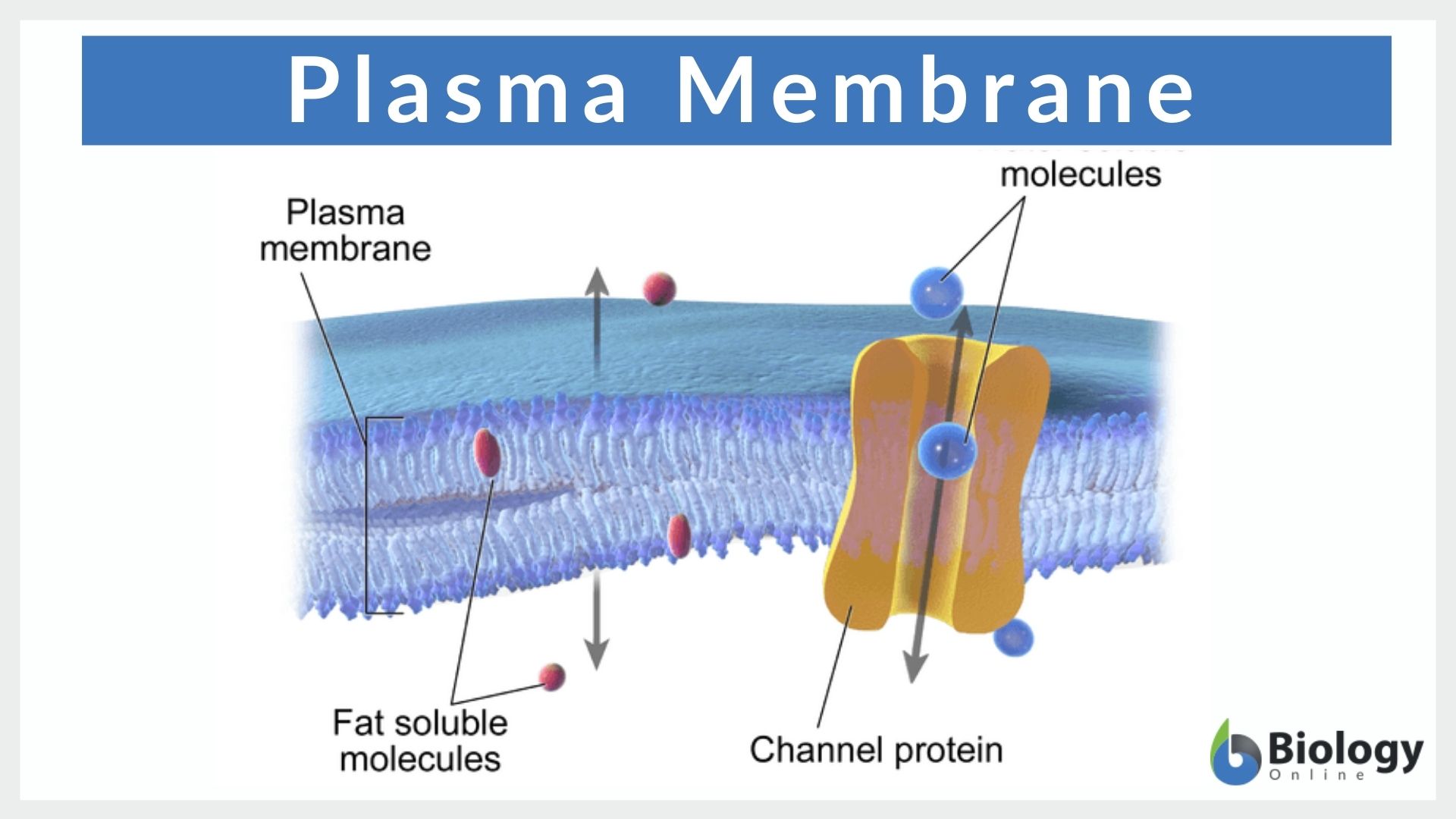
In hydrophobicity scales, the more hydrophobic amino acids have the highest positive values while the more hydrophilic ones have the highest negative values. Hydrophobicity scales are values that define the relative hydrophobicity or hydrophilicity of amino acid residues.
